Fusion reactors rely on high-purity lithium to generate tritium, the fuel that sustains deuterium–tritium (D-T) reactions. Owing to the scarcity of tritium, lithium breeder blankets (Figure 1) are employed to surround the plasma and capture high-energy neutrons. This converts lithium into tritium, enabling a self-sufficient fuel cycle. Beyond its use in breeder blankets, lithium also serves as a coolant and protective buffer, reducing heat and radiation damage to reactor structures. The required concentration, isotopic composition, and blanket geometry vary with design, with the aim to maximize tritium production and reactor efficiency. High-purity lithium is therefore a critical factor for reliable fusion performance.
The primary D–T fusion reaction is given by Eq. 1:
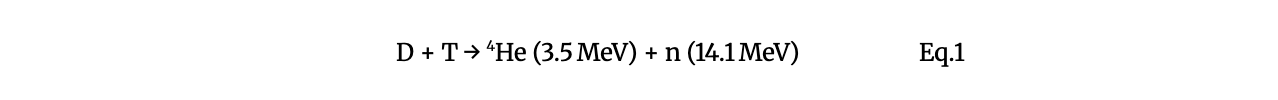
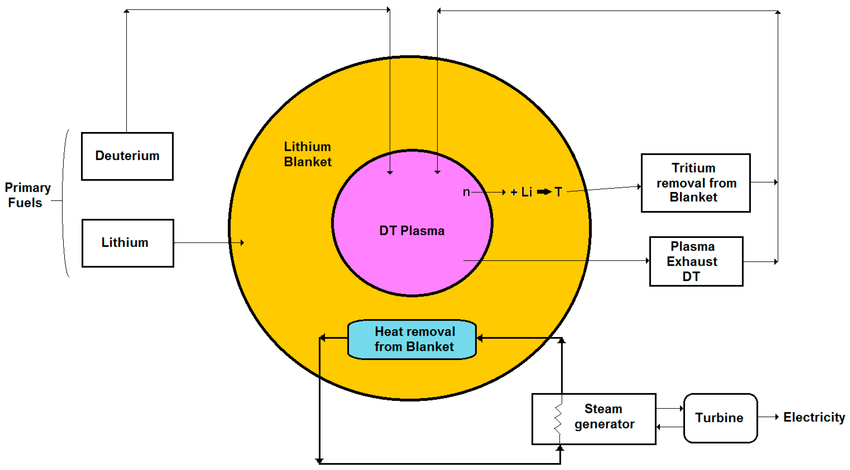
Figure 1: Schematic of a fusion power plant highlighting the plasma core, lithium breeder blanket, and heat extraction systems.
Even trace impurities in lithium can significantly reduce tritium production and compromise machine efficiency. Unlike battery-grade lithium, which tolerates a variety of chemical contaminants, fusion-grade lithium must meet stringent nuclear-level standards, although these standards are still evolving. Understanding which impurities matter, and why, is critical for operational success.
Chemical impurities such as oxygen and nitrogen directly interfere with lithium chemistry and plasma stability. For oxygen, concentrations must be kept below a critical threshold of 100 ppm to prevent accelerated corrosion of certain refractory metals, such as tantalum. Failure to do so leads to oxide precipitation, poorer heat transfer, and higher maintenance costs [1]. Nitrogen, meanwhile, acts as a strong radiative impurity inside the plasma core, cooling the plasma and diluting the fusion fuel [2]. This reduces net power output, and operators must install advanced diagnostics and control systems to monitor impurity levels and maintain stable plasma conditions.
Trace metals are another significant concern in fusion-grade lithium, including iron, chromium, nickel, and copper. They can be present naturally in lithium sources or introduced during production and handling. Even at very low concentrations, these metals absorb neutrons, which can reduce the tritium breeding ratio (TBR) and limit the reactor’s fuel self-sufficiency. In addition, trace metals can chemically interact with lithium or other impurities, subtly affecting chemical stability and thermal properties, which can further decrease tritium production efficiency and overall reactor performance. For these reasons, careful purification and continuous monitoring of trace metal concentrations are essential to maintain operational reliability and ensure the long-term performance of the breeder blanket
In addition to metals, lithium can contain trace amounts of hydrogen isotopes, hydrogen, deuterium, and tritium, introduced naturally or through contact with moisture during processing. Due to lithium’s strong chemical affinity for these isotopes, it readily forms stable hydrides (LiH, LiD, LiT), which temporarily trap fuel and complicate tritium extraction [3]. In high-performance facilities like International Fusion Materials Irradiation Facility (IFMIF), strict impurity limits are maintained, less than 10 wppm for deuterium and less than 1 wppm for tritium, to ensure lithium flow stability & safety in a neutron source [4]. Despite these limits, hydride formation can still occur, requiring careful management during tritium extraction using methods such as electrolysis or yttrium hot traps [4]. Maintaining both low hydrogen isotope levels and efficient extraction systems is therefore essential to preserve lithium’s chemical stability and achieve self-sufficient fuel operation in fusion reactors.
These impurity pathways highlight how fusion-grade lithium differs fundamentally from commercial battery-grade lithium, requiring not only advanced separation and monitoring technologies but also new certification frameworks to ensure nuclear performance standards are met. Without such measures, lithium impurities can erode reactor reliability, increase operating costs, and undermine the economic viability of fusion power.
References
[1] R. L. Klueh, Metallurgical transactions, 1974, 5, 875–879.
[2]M. Oberkofler, D. Alegre, F. Aumayr, S. Brezinsek, T. Dittmar, K. Dobes, D. Douai, A. Drenik, M. Köppen, U. Kruezi, Ch. Linsmeier, C. P. Lungu, M. Mozetič, C. Porosnicu, V. Rohde, S. G. Romanelli, the ASDEX Upgrade Team and JET EFDA Contributors, Plasma-wall interactions with nitrogen seeding in all-metal fusion devices: formation of nitrides and ammonia, in Proc. 28th Symposium on Fusion Technology (SOFT), San Sebastian, Spain, 29 Sept.–3 Oct. 2014, preprint.
[3]D. Zheng, Z.-X. Shen, M. Chen, X. Ren and L. He, Journal of Nuclear Materials, 2021, 543, 152542.
[4] K. Esaki, K. Hiyane, S. Fukada, E. Wakai, Y. Ito, F. Nitti and JAEA IFMIF group, J. Plasma Fusion Res. SERIES, 2015, 11, 36.
Figure 1: S. Tosti and A. Pozio, Membranes, 2018, 8, 96.
